Abstract

BACKGROUND: Genomic aberrations, IGHV mutation status and recurrent mutations in genes such as TP53 are established prognostic factors in CLL in the context of chemoimmunotherapy (CIT). For time limited treatment with venetoclax in combination with Rituximab (RVe), Obinutuzumab (GVe) and Obinutuzumab and Ibrutinib (GIVe) the role of these factors is less-well established.
METHODS: The phase 3 GAIA/CLL13 trial compared 3 different time-limited venetoclax-based combinations against CIT in fit, treatment-naïve patients with CLL without del(17p) or TP53 mutations detected via Sanger sequencing. Patients were randomized to receive CIT (FCR for patients ≤65y; BR for patients >65y), RVe, GVe or GIVe. Co-primary endpoints were PFS and MRD in peripheral blood at month 15 via 4-colour flow.
Genomic aberrations were assessed at enrolment by FISH, IGHV by sequencing with a threshold of 98% homology and mutations via amplicon-based targeted NGS for TP53, NOTCH1, SF3B1, MYD88, KRAS, NRAS, BRAF, EGR2, NFKBIE, RPS15, XPO1 and BIRC3 in 913 of 926 patients (98.6%) of the full trial cohort.
RESULTS: The incidences of genomic aberrations based on the hierarchical model were del(11q) 17.5%, +12 16.2%, mono-allelic del(13q) 44.6% and normal karyotype 21.7%. IGHV was unmutated (U-IGHV) in 55.9% and mutated (M-IGHV) in 41.0% of patients (ITT). Subset#2 accounted for 50 patients equally distributed among U-IGHV and M-IGHV. The incidence of gene mutations was SF3B1 22.6%, NOTCH1 20.5% (exon 34 and 3'UTR), NFKBIE 13.6%, BIRC3 11.8%, BRAF/KRAS together 9.6%, XPO1 7.9%, EGR2 5.6%, RPS15 5.5% and MYD88 4.2%. Minor mutations with a variant allele fraction (VAF) of 1.5% to 10% were frequent in BIRC3 and NFKBIE (59.6% and 56.0% of all variants, respectively), while minor mutations in TP53 not previously detected by Sanger sequencing were rare (2.0%).
Undetectable MRD (uMRD) was not associated with any genetic subgroup in the pooled analysis of GVe/RVe/GIVe. Single arm analysis revealed NOTCH1 mutation associated with lower rate of uMRD with GVe (74.4% vs 88.6%, p=0.02) and higher uMRD rate in +12 cases with RVe (73.5% vs. 52.2% in del(13q), p=0.03). With CIT a significantly lower uMRD rate was found in U-IGHV (42.0% vs. 66.3% in M-IGHV, p<0.01) and del(11q) (39.0% vs. 58.4% in del(13q), p=0.04) while mutated MYD88 associated with a higher uMRD rate (92.3% vs 50.2%, p=0.02).
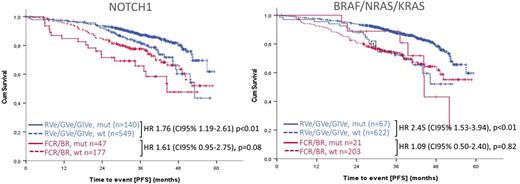
At a median follow-up of 38.8 months, there were 188 PFS events in the ITT population. For CIT U-IGHV (HR 4.47, p<0.01), del(11q) (HR 2.68, p<0.01) and mutated NOTCH1 with VAF>10% (HR 2.01, p=0.01) associated significantly with shorter PFS in univariate analysis. In the combined analysis for RVe/GVe/GIVe U-IGHV (HR 2.29, p<0.01), mutations in NOTCH1 (HR 1.76, p<0.01) and BRAF/RAS (HR 2.45, p<0.01) were associated with significantly shorter PFS and del(13q) (HR 0.67, p=0.03) with longer PFS. A separate analysis for each venetoclax arm identified mutated NOTCH1 (GVe: HR 2.2 p=0.04 and GIVe: HR 2.2, p=0.05), BIRC3 (GIVe: HR 3.6 p<0.01), BRAF/KRAS/NRAS (RVe: HR 3.6, p<0.01 and GIVe: HR 3.7, p<0.01) NFKBIE (GVe: HR 3.0, p<0.01), del(13q) (GVe: HR 0.4, p=0.01) and U-IGHV (GVe: HR 2.6, p=0.03 and GIVe: HR 2.7, p=0.03) as prognostic factors for PFS. Subset 2 was not associated with shorter PFS, also not among M-IGHV patients.
In multivariable analysis including all clinical, laboratory and genetic markers significantly associated with PFS, treatment with GVe and GIVe were independent favorable factors (HR 0.42 and 0.33, respectively, both p<0.01) and U-IGHV (HR 2.43, p<0.01), and mutated NOTCH1 (HR 1.46, p=0.03) were confirmed as independent prognostic factors. In a separate multivariable analysis only for CIT, del(11q) (HR 1.89, p=0.03), U-IGHV (HR 3.08, p<0.01), and mutated NOTCH1 >10% VAF (HR 2.12, p=0.01) were verified as independent adverse genetic risk factors. For the combined RVe/GVe/GIVe group, mutated BRAF/RAS (HR 1.87, p=0.01), U-IGHV (HR 1.85, p<0.01), and mutated NOTCH1 (HR 1.54, p=0.04) were confirmed as independent adverse prognostic factors.
CONCLUSIONS: Early results from CLL13/GAIA show association of gene mutations with lower rate for uMRD and shorter PFS after CIT or venetoclax-based therapy. IGHV and NOTCH1 mutation retain their prognostic role with both treatment types. Mutations in the RAF/RAS pathway (9.6% overall) were associated with shorter PFS with venetoclax-based therapy, while with CIT an impact on outcome is not apparent at present.
Disclosures
Tausch:Abbvie: Consultancy, Research Funding, Speakers Bureau; Roche: Consultancy, Research Funding, Speakers Bureau; Janssen-Cilag: Honoraria; BeiGene: Honoraria; AstraZeneca: Honoraria, Speakers Bureau. Schneider:AbbVie, AstraZeneca: Honoraria, Speakers Bureau. Robrecht:AstraZeneca: Honoraria. Staber:roche: Consultancy, Honoraria, Research Funding; amgen: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; Morphosys: Consultancy, Honoraria; CTI: Consultancy, Honoraria; BMS: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Beigene: Consultancy, Honoraria; Lilly: Consultancy, Honoraria. Tadmor:Janssen: Research Funding; AbbVie, Roche, Novartis, Sanofi, Takeda, Janssen, Pfizer: Consultancy, Honoraria, Speakers Bureau. Levin:AbbVie, Roche and Janssen: Other: Travel expenses. Janssens:Genmab: Current Employment; Abbvie: Consultancy, Other: Travel Grants, Speakers Bureau; Amgen: Consultancy, Other: travel grants, Speakers Bureau; Astra-Zeneca: Consultancy, Speakers Bureau; Beigene: Consultancy, Speakers Bureau; Janssen: Consultancy, Speakers Bureau; Incyte: Consultancy, Speakers Bureau; Novartis: Consultancy, Speakers Bureau; Sobi: Consultancy, Speakers Bureau; Sanofi Genzyme: Consultancy, Speakers Bureau; Roche: Consultancy; Celgene: Other: travel grants. Noesslinger:abbvie: Consultancy; celgene: Consultancy; janssen: Consultancy; gilead: Consultancy; roche: Consultancy; beigene: Consultancy; astrazeneca: Consultancy. Baumann:AbbVie, AstraZeneca, BMS/Celgene, Jannsen, Novartis, Pfizer, Roche: Consultancy. Wendtner:Hoffmann-La Roche, Cilag-Janssen, AbbVie: Consultancy, Research Funding. Kreuzer:AbbVie, Amgen, Celgene, F. Hoffmann-LaRoche, Gilead, Janssen-Cilag, Mundipharma: Consultancy, Honoraria, Research Funding, Speakers Bureau. Fink:Abbvie: Other: Travel Grants; Celegene, AstraZeneca: Research Funding. Fischer:AbbVie: Honoraria; Astra Zeneca: Membership on an entity's Board of Directors or advisory committees; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Support. Kater:BMS: Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees, Other: speakers fee, Patents & Royalties: pending, Research Funding; Roche/genentech: Membership on an entity's Board of Directors or advisory committees; LAVA: Membership on an entity's Board of Directors or advisory committees, Patents & Royalties: pending; Astra Zeneca: Membership on an entity's Board of Directors or advisory committees, Other: speakers fee, Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees, Other: speakers fee, Research Funding. Niemann:Takeda: Consultancy; CSL Behring: Consultancy; Octapharma: Consultancy, Research Funding; Janssen: Consultancy; AstraZeneca: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding; Beigene: Consultancy; Genmab: Consultancy. Hallek:Gilead: Consultancy, Honoraria; Abbvie, AstraZeneca: Consultancy, Honoraria, Research Funding; Roche, Janssen: Honoraria, Research Funding; Bristol Myers Squibb: Consultancy, Honoraria. Eichhorst:Janssen, AbbVie, Lilly, AstraZeneca, BeiGene, MSD: Consultancy; Janssen, Roche, AbbVie, BeiGene, AstraZeneca: Research Funding; Janssen, Roche, AbbVie, BeiGene, AstraZeneca, MSD: Speakers Bureau; Beigene: Other: Travel Support. Stilgenbauer:Beigene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; AbbVIe: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Acerta: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; AstraZeneca: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Genentech: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; GSK: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Hoffmann-La Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Incyte: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Infinity: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Pharmacyclics: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Sunesis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Veristem: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau.
OffLabel Disclosure:
RVe and GIVe are therapies not approved in first line CLL
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal